The Medtronic MiniMedTM 670G system has received Health Canada license with sales expected to begin in Fall 2018.
Read on to learn a bit more about the product and to find out if it’s in the running for your next pump.

I’M JUST GETTING TO KNOW MEDTRONIC. CAN YOU TELL ME A BIT MORE ABOUT THE COMPANY?
Medtronic was founded way back in 1949 by Earl Bakken and his brother-in-law, Palmer Hermundslie. The company became pioneers of many medical devices, including the first battery-powered pacemaker. Medtronic is now the world’s largest medical technology company. The first insulin pump came to market from Medtronic in 1983 – MiniMed® 502. It wasn’t until 1999 that the first Continuous Glucose Monitor was introduced. Today, Medtronic has several pumps on the market. In the USA, the first closed-loop hybrid system combining CGM and pumping technology was launched in 2017. Learn more about Medtronic’s innovation milestones, here.
HOW IS MEDTRONIC ENGAGING WITH THE TYPE 1 DIABETES COMMUNITY?
It can actually be quite tricky to find more about what companies are doing to support the community that they’re serving (that’s us!), outside of providing products and services. We believe this is one of the most important questions we can ask. We’re grateful to the companies that are giving back and are proud to see the other work that Medtronic is doing in the community. We wanted to share what we could find with you!
We asked Medtronic directly what they had going on in the community: “Medtronic is a company driven by its Mission and works towards creating a better, healthier world for patients and their communities. Within Diabetes, Medtronic Canada engages with many patient communities across Canada that focuses on education, research, advocacy, support and a commitment to helping improve the lives of those touched by diabetes.”
Medtronic is a Partner of Connected in Motion and has been a supporter of what we do since Day 1. Check out what programs they are supporting on our Partners Page.
WHAT’S NEW WITH THE 670G?
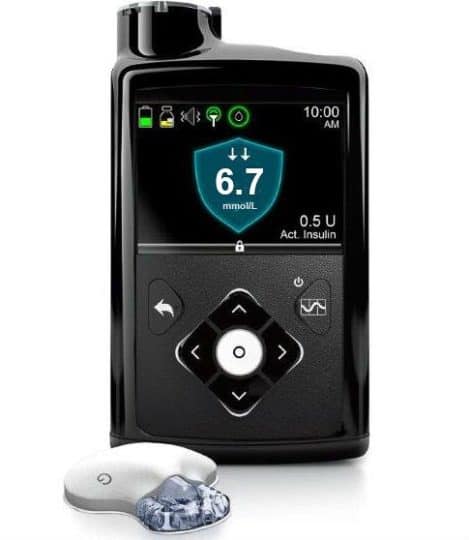
The one big difference when comparing the 670G and the 630G is the hybrid closed loop function.
Hybrid-Closed Loop: This is the new and perhaps most exciting part. It functions by making adjustments to the BASAL insulin based on data received from the linked Guardian Sensor 3 glucose transmitter. Basal changes are made every 5 minutes based on that sensor data, carb data entered, and insulin on board. The algorithm is also ‘smart’ and looks at your trends and BG reactions from the past 6 days to inform basal adjustments.
The algorithm is quite conservative. The pump will be hard-set with a target BG of 6.7 mmol/L (*assuming the same settings as in the US which will hard-set at 120 mg/dL). While this is a great first step in the hybrid-closed loop world, it may be too conservative for those wanting very tight ranges, women who are pregnant, certain athletes, etc.
The pump can also function out of Auto-Mode. (Auto-mode being when basal adjustments are being automatically made.) In fact, you’ll be kicked out of auto-mode into safe-mode for a variety of reasons (sensor is in warm-up mode or requires calibration, occlusion is detected, signal loss, etc.). You may also just choose to stop using auto-mode for a period of time. Know that it’s an option.
THE DETAILS – GUARDIAN SENSOR 3
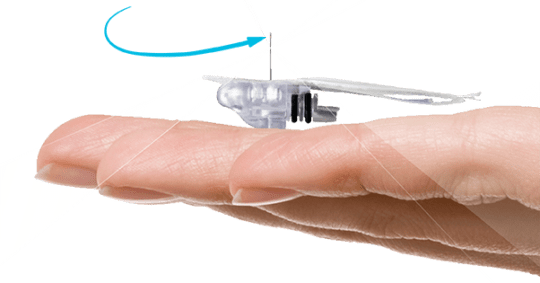
The enhanced Enlite was released in Canada in 2014, a second version of the original Enlite sensor. With the launch of the 670g comes the launch of Medtronic’s newest sensor, the Guardian Sensor 3. There are several upgrades to this sensor from the original version, listed below. The Guardian Sensor 3 is also the part of the 670G system that really makes this pump special:
- Sensor size: The actual part that is inside your body sensing is 80% smaller than previous Enlites
- Consistent: The new design allows more consistent reading over the wear of the sensor
- Connection: The wireless connection allows for better communication between the sensor and the pump for fewer lost signal alarms
- Adhesive: The adhesive has been updated to cause less skin irritation.
OTHER DETAILS – MINIMED 670G
- Reservoir Size: Can hold 300 units of Rapid-Acting Insulin
- Site Specifics: Works with Medtronic’s line of pump sites – Quick-set, Silhouette, Mio, Mio 30, and Sure-T.
- Waterproof: Up to 3.6m for 24 hours
- Bolus Wizard: Built-in bolus calculator that considers active insulin, carbs and BG to help determine precise bolus amounts
- Colour Home Screen: Home screen shows active insulin, CGM reading (if using CGM) OR last BG readings entered in past 12 minutes, bolus and basal options
- Basal Patterns: Ability to save 8 basal patterns (think, sick day, aerobic workout day, travel day, ‘time of the month’ day… the list goes on!)
- Preset Bolus: You can save preset boluses. Do you always bolus the same for breakfast? Save that bolus in your pump for easy access
- Event Markers & Reminders: You can tag events on your pump to show up in your records – exercise, feeling sick, stressed, etc. You can also have the pump send you reminders: Don’t forget to set temp basal, check BG 2 hours after meal, etc.
I REALLY VALUE THE EXPERIENCES OF THE COMMUNITY. ARE THERE ANY REVIEWS OF THE 670G OUT THERE YOU COULD SUGGEST READING?
Sure! You think just like us. Check these out these reviews from the US – note, the BG measurement they reference is different from what we use here in Canada. Don’t worry. People are generally happy with a BG of 100 in the US!:
Detailed Review of the MiniMed 670g by Integrated Diabetes Solutions
Review: “World’s First Artificial Pancreas” by dLife
What it’s like to use a 670g by A Sweet Life
Getting personal with Medtronic’s Minimed 670G by Healthline
Disclaimer: Although this is not paid content, Medtronic, Omnipod, Tandem and Dexcom are national partners of Connected in Motion. Our views may be biased because of these partnerships, but we still thought this information was helpful for the community.





